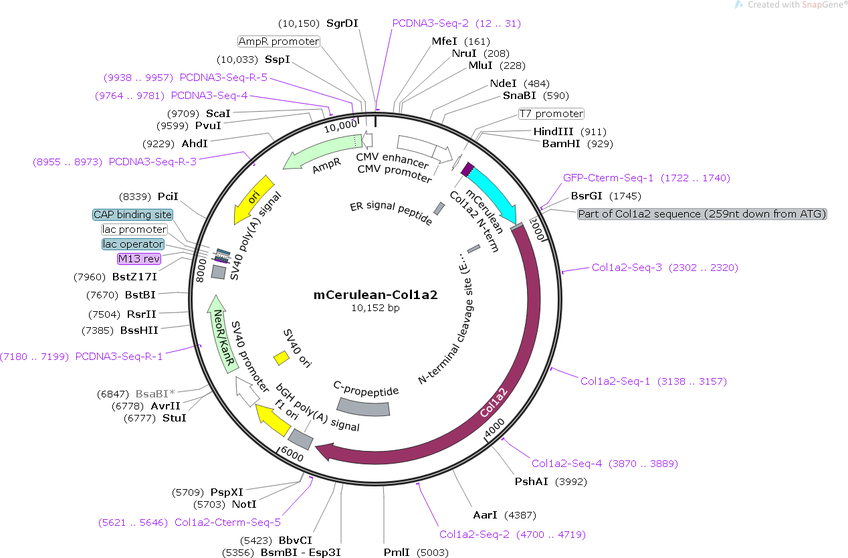
mCerulean-proα2(I)
(Plasmid
#119836)
-
PurposeExpresses mouse Type I procollagen α2 chain (Col1a2) with Gly610Cys mutation and mCerulean between signal sequence and exon 6
-
Depositing Lab
-
Sequence Information
Ordering
| Item | Catalog # | Description | Quantity | Price (USD) | |
|---|---|---|---|---|---|
| Plasmid | 119836 | Standard format: Plasmid sent in bacteria as agar stab | 1 | $85 | |
Backbone
-
Vector backbonepcDNA3.1
- Backbone size w/o insert (bp) 5400
- Total vector size (bp) 10153
-
Vector typeMammalian Expression
-
Selectable markersNeomycin (select with G418)
Growth in Bacteria
-
Bacterial Resistance(s)Ampicillin, 100 μg/mL
-
Growth Temperature37°C
-
Growth Strain(s)DH5alpha
-
Growth instructionsOriginally grown in One Shot TOP10 Chemically competent E. coli
-
Copy numberUnknown
Gene/Insert
-
Gene/Insert nameType I procollagen α2 chain
-
Alt nameCol1a2
-
SpeciesM. musculus (mouse)
-
Insert Size (bp)4737
-
MutationCol1a2 exons 2-5 replaced by fluorescent tag
-
GenBank IDNM_007743
-
Entrez GeneCol1a2 (a.k.a. Col1a-2, Cola-2, Cola2, oim)
- Promoter CMV
-
Tag
/ Fusion Protein
- mCerulean (N terminal on insert)
Cloning Information
- Cloning method Restriction Enzyme
- 5′ cloning site HindIII, BamHI (not destroyed)
- 3′ cloning site NotI (not destroyed)
- 5′ sequencing primer CGC AAA TGG GCG GTA GGC GTG
- 3′ sequencing primer TAG AAG GCA CAG TCG AGG (Common Sequencing Primers)
Resource Information
-
Supplemental Documents
Terms and Licenses
-
Academic/Nonprofit Terms
-
Industry Terms
- Not Available to Industry
Trademarks:
- Zeocin® is an InvivoGen trademark.
Depositor Comments
These mouse type I procollagen plasmids were designed for immortalized and primary murine osteoblasts and fibroblasts. Their expression in other cells might cause significant disruption of assembly, folding and trafficking of type I procollagen, resulting in artifacts. Even in osteoblasts and fibroblasts a fraction of transfected chains, particularly proα2, might be trafficked/degraded as monomers instead of being integrated into procollagen heterotrimers. In our experience, the best evidence of normal behavior of the transfected chains is the appearance of fluorescent extracellular collagen fibers ~ 12 h after transfection. Fluorescent molecules should be incorporated into fibers even when the transfected chains contain the N-propeptide cleavage site, because the cleavage is highly conformation sensitive and always incomplete.
We recommend following these guidelines when using the plasmids
1) Only mouse cells that have high expression of endogenous type I procollagen are suitable for studies of physiologically-relevant processes with these constructs.
2) At least 100 μM ascorbic acid at and after transfection is required to ensure proper procollagen folding (for some cells, preincubation with ascorbic acid before transfection might be needed).
3) Optimization of transfection efficiency is needed for all cell types.
4) Experiments beyond 24h after transfection are not recommended, to avoid excessive accumulation of aggregates and cell malfunction caused by increased procollagen synthesis.
5) Lack of extracellular fluorescent fibers 12-24 h after transfection indicates improper procollagen synthesis/trafficking or cellular malfunction.
6) Excessive expression of transfected chains might cause rapid accumulation of large procollagen aggregates in the ER, resulting in cellular malfunction and death.
Contact information: Shakib Omari ([email protected]) Sergey Leikin ([email protected])
These plasmids were created by your colleagues. Please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
-
For your Materials & Methods section:
mCerulean-proα2(I) was a gift from Sergey Leikin (Addgene plasmid # 119836 ; http://n2t.net/addgene:119836 ; RRID:Addgene_119836) -
For your References section:
Noncanonical autophagy at ER exit sites regulates procollagen turnover. Omari S, Makareeva E, Roberts-Pilgrim A, Mirigian L, Jarnik M, Ott C, Lippincott-Schwartz J, Leikin S. Proc Natl Acad Sci U S A. 2018 Oct 23;115(43):E10099-E10108. doi: 10.1073/pnas.1814552115. Epub 2018 Oct 4. 10.1073/pnas.1814552115 PubMed 30287488